A2AR, ACTIVE
Recombinant human Adenosine A2A receptor full-length, wildtype sequence N-terminus Strep tag II, 8xHis-tag, TEV protease cleavage site
Biological information
Background
Adenosine receptor A2A aids in the discovery, development, and validation of new molecules as well as therapeutic antibodies. The receptor's function is facilitated through G proteins, which, in turn, stimulate adenylyl cyclase. This membrane protein is ideal for CNS neuroprotection in order to develop new treatments for insomnia, pain, depression, Alzheimer and Parkinson’s diseases. This class A GPCR is involved in the regulation of myocardial blood flow/hypertension and has a N-terminal Step tag II and an 8X His tag followed by a TEV protease cleavage site. Adenosine receptor A2A is a powerful therapeutic target especially found in the treatment of such conditions as insomnia, pain, depression, and Parkinson’s disease. Our A2AR protein is produced in a eukaryotic system with the proper post-translational modifications (glycosylation). Our Adenosine receptor A2A also maintain its structural and functional integrity and is purified and stabilized to full length and wild-type (native) proteins.
Target class
GPCR
Family
Class A GPCR
Accession number
P29274
Target Name
A2AR, ACTIVE
Target Alias
Origin
Human
Theori. MW
47.7kDa
Affinity tag
Strep tag II, 8xHis-tag
Product specifications
Expression system
Expressed in baculovirus in Sf9 insect cells
Purity
Purity >90% by SDS-PAGE with Stain-FreeTM detection and western blotting with an HRP-coupled monoclonal anti-ICL3-A2A
Purification method
Immobilized Metal Affinity Chromatography, StrepTactin XT affinity chromatography
Sample Buffer
Protein in 25 mM Na2HPO4/NaH2PO4 pH 8.0, 150 mM NaCl, 0.05%-0.005% DDM-CHS.
Specified activity
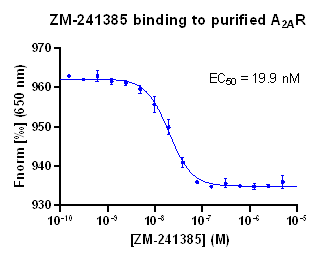
Confirmed by MicroScale Thermophoresis ligand binding assay and Radio ligand binding assay
Application
For Research Only
Storage conditions
Store at -80°C
Usage disclaimer
For Research Only
Chemical data
Background
Adenosine receptor A2A aids in the discovery, development, and validation of new molecules as well as therapeutic antibodies. The receptor's function is facilitated through G proteins, which, in turn, stimulate adenylyl cyclase. This membrane protein is ideal for CNS neuroprotection in order to develop new treatments for insomnia, pain, depression, Alzheimer and Parkinson’s diseases. This class A GPCR is involved in the regulation of myocardial blood flow/hypertension and has a N-terminal Step tag II and an 8X His tag followed by a TEV protease cleavage site. Adenosine receptor A2A is a powerful therapeutic target especially found in the treatment of such conditions as insomnia, pain, depression, and Parkinson’s disease. Our A2AR protein is produced in a eukaryotic system with the proper post-translational modifications (glycosylation). Our Adenosine receptor A2A also maintain its structural and functional integrity and is purified and stabilized to full length and wild-type (native) proteins.
Compound name
GPCR
Catalog number
41-G101A-10UG
Molecular formula
CAS
MW
Ka
Percent composition
Product specifications
Physical state
Purity (HPLC 214nm)
Retention time (RP18 HPLC)
CMC
Exact mass
Stability
For Research Only
Solubility structure
Datasheets
41-G101A
Datasheet